
In order for a reaction to occur, one of the products is usually a solid precipitate, a gas, or a molecular compound such as water. Double-replacement reactions generally occur between substances in aqueous solution. Which of the following is a precipitation reaction Zn(s) + 2 AgNO3(aq) 2 Ag(s) + Zn(NO3)2(aq). Hg2I2 Na2SO4 Na2CO3 CaS All of these compounds are soluble in water. Calculate the amount of the second reactant, Na2CO3, needed to make a precipitate of calcium carbonate using stoichiometry. Which of the following pairs of aqueous solutions will form a precipitate when mixed NH4NO3 + K2CO3 Hg2(NO3)2 + NaBr KCl + Li3PO4 AgC2H3O2 + Cu(NO3). 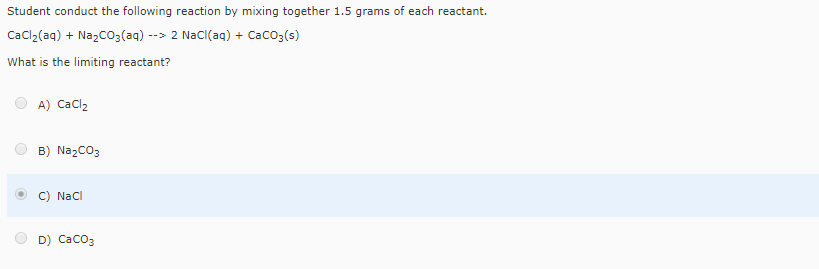
Add 25 mL of distilled water to the beaker containing CaCl2♲H2O and stir the mixture. Na2CO3(aq) + CaCl2(aq) CaCO3(s) + 2NaCl In ideal conditions, the percent yield of the reaction must be 100. 
The general equation for double displacement reaction is:ĭouble displacement reactions can be further classified as neutralization, precipitation and gas formation reactions. Weigh out 1.0 gram of CaCl2♲H2O on a digital scale and place it in a 100 mL beaker. Na2CO3(aq) 2Na+ (aq) + CO2 3(aq) Likewise, the calcium chloride solution. You can thus say that the sodium carbonate solution will contain sodium cations, Na+, and carbonate anions, CO2 3. Therefore, when aqueous solutions of calcium chloride and of sodium carbonate are mixed, a double displacement reaction takes place.ĭouble displacement reactions take place in aqueous solutions in which the ions precipitate and there is an exchange of ions. Sodium carbonate, Na2CO3, and calcium chloride, CaCl2, are soluble ionic compounds that dissociate in aqueous solution to form cations and anions. Hence, we can say that the above reaction is a double displacement reaction. Try to identify the name of the reaction taking place here.Ĭalcium chloride $ \left( $īy looking at the reaction we can say that in the above reaction the cationic part of the reactant are getting displaced by each other and are getting exchanged, which is resulting in formation of new products. (10 pts) For each mixture below, state if a precipitate will form or not. a) Add CaCl2 to Na2CO3 b) Add Ca(OH)2 to Na2SO4 c) Add Ca(OH)2 to KCl 3. CaCl2(aq) + Na2CO3(aq) -> CaCO3(s) + 2 NaCl(aq) What is the complete ionic. When there is formation of products, one of them will get precipitated and the other remains. If a precipitate forms, write a BALANCED equation for the reaction and CIRCLE the compound(s) which is the precipitate. When you mix calcium chloride with sodium carbonate, the following reaction occurs. Hint: Write the formula of the names given and figure out the reaction taking place between the two.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
